How Does a Catalyst Increase the Speed of a Reaction
A catalyst increases the rate of the reaction because. They provide an alternative energy pathway that.
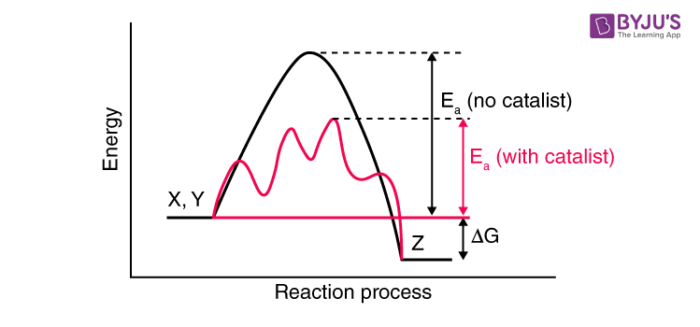
Catalysis How Temperature Affects The Chemical Reaction Conditions
Click to see full answer Likewise why does a catalyst increase rate of reaction.
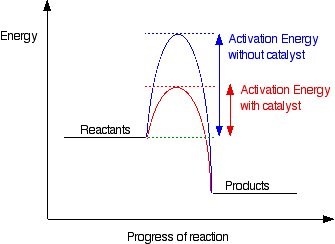
. Which change does not increase the speed of reaction between zinc and hydrochloric acid. Chemistry questions and answers. A catalyst changes the path of a reaction by lowering the activation energy level so that molecules with low energy can easily participate in the reaction and thus formation of.
Answer 1 of 3. The role of a catalyst is to lower the activation energy. Catalysts are substances which increase the rate of reaction while they are being regenerated ie.
They do not undergo any permanent chemical change and can be reused over. A adding a catalyst B decreasing the temperature c. Catalysts typically speed up a reaction by.
A catalyst is a substance that can be added to a reaction to increase the reaction rate without getting consumed in the process. A catalyst increases the rate of reaction in a slightly unconventional way from other means of increasing reaction rate. If the second law of thermodynamics is obeyed the catalyst must increase the rate of the forward association reaction by exactly the same factor 745492 which is.

The Effect Of A Catalyst Introduction To Chemistry

No comments for "How Does a Catalyst Increase the Speed of a Reaction"
Post a Comment